Amyotrophic lateral sclerosis (ALS) is still incurable. Although different therapies can affect the health and survival of patients. Our aim is to evaluate the effect of umbilical mesenchymal stem cells administrated intrathecally to patients with amyotrophic lateral sclerosis on disability development and survival.
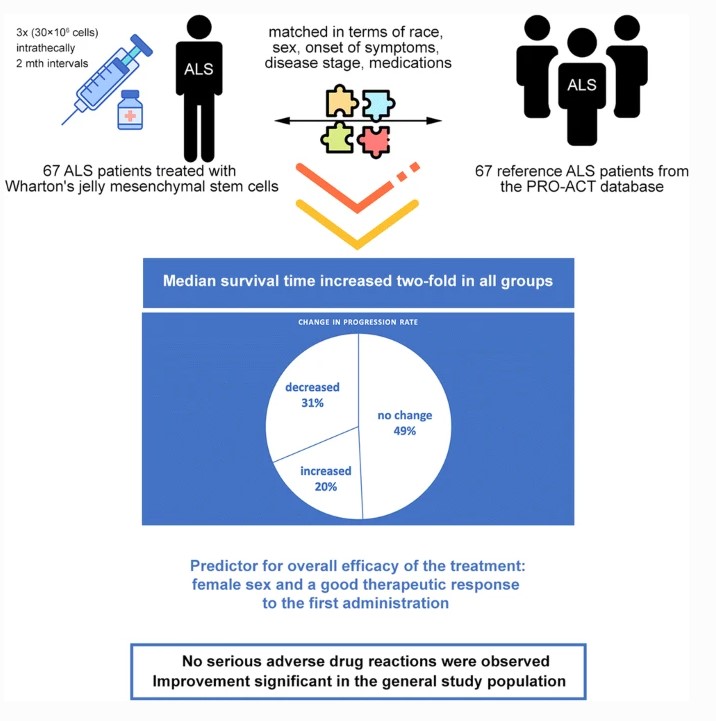
This case-control study involved 67 patients treated with Wharton’s jelly mesenchymal stem cells (WJ-MSC). The treated patients were paired with 67 reference patients from the PRO-ACT database which contains patient records from 23 ALS clinical studies (phase 2/3). Patients in the treatment and reference groups were fully matched in terms of race, sex, onset of symptoms (bulbar/spinal), FT9 disease stage at the beginning of therapy and concomitant amyotrophic lateral sclerosis medications. Progression rates prior to treatment varied within a range of ± 0.5 points. All patients received three intrathecal injections of Wharton’s jelly-derived mesenchymal stem cells every two months at a dose of 30 × 106 cells. Patients were assessed using the ALSFRS-R scale. Survival times were followed-up until March 2020.
Median survival time increased two-fold in all groups. In terms of progression, three response types measured in ALSFRS-R were observed: decreased progression rate (n = 21, 31.3%), no change in progression rate (n = 33, 49.3%) and increased progression rate (n = 13, 19.4%). Risk-benefit ratios were favorable in all groups. No serious adverse drug reactions were observed.
Wharton’s jelly-derived mesenchymal stem cells therapy is safe and effective in some ALS patients, regardless of the clinical features and demographic factors excluding sex. The female sex and a good therapeutic response to the first administration are significant predictors of efficacy following further administrations.
Keeping in mind the study limitation, our results suggest that treatment with MSC substantially reduces the rate of progression and yields a twofold extension of survival in those who achieved a positive response to the first administration in comparison to the reference subject.
These HR values were even better than those achieved with the registered drug, edaravone. Demographic factors have a certain impact on the risk-benefit ratio, so they should be discussed with the patient before the start of therapy. MSC administration was absolutely safe. Eligibility for the treatment should be confirmed following first administration based on the progression rate in comparison with at least one reference subject (more would be beneficial). Future studies should focus on confirming these observations in double-blind, controlled studies, optimally with a three-arm design to investigate additive or synergic effects with edaravone.
CLINICAL STUDY link.springer.com/article/10.1007/s12015-020-10016-7?elqTrackId=e04512242c98421ba643066ab0c83a41